
Left-to-right shunt for Heart Failure
Despite advances over the past decade, Heart Failure (HF) remains a significant healthcare and economic burden. According to the American Heart Association (AHA), 6.2 million Americans aged 20 and older suffer from HF and the number is expected to increase by almost 50% by 2030. Worldwide, HF affects over 30 million people.
The lifetime risk of HF increases with age, and over 50% of hospitalizations of persons aged 65 or older are attributable to HF. In the United States, the total HF-related costs were estimated to USD 30.7 billion in 2012. Key cost-drivers include the re-hospitalization rates of over 50%, treatment costs, and the fact that no effective treatments are available. Projections suggest that by 2030, the total HF cost will increase to USD 69.8 billion. Heart failure (HF) is a serious condition characterized by the heart’s inability to pump an adequate supply of blood to the body.
Symptoms include fatigue, palpitations, and exertional dyspnea that may be accompanied by specific signs such as elevated jugular venous pressure, pulmonary crackles, and peripheral edema. HF may result from disorders of the pericardium, myocardium, endocardium, heart valves, great vessels, or specific metabolic abnormalities. These disorders affect the structure or function of the heart which results in reduced cardiac output and/or elevated intracardiac pressures at rest or during stress. Left untreated, symptoms gradually worsen, resulting in increased morbidity, clinically driven hospitalizations, and higher mortality.
Ejection fraction (EF) is considered an important factor for the classification of HF patients and left ventricular ejection fraction (LVEF) is used to group HF patients into three subtypes:
- Heart failure with preserved ejection fraction (HFpEF): patients with normal LVEF (preserved LVEF ≥ 50%) and elevated levels of natriuretic peptides (B-type natriuretic peptide (BNP) > 35 pg/ml and/or N-terminal pro-B type
natriuretic peptide (NT-proBNP) >125 pg/mL. - Heart failure with mid-range ejection fraction (HFmrEF): patients with midrange LVEF (40–49%) and elevated levels of BNP > 35 pg/ml and/or NTproBNP > 125 pg/mL.
- Heart failure with reduced ejection fraction (HFrEF): Patients with
markedly reduced LVEF < 40% (HFrEF).HFpEF and HFrEF patients have different profiles. Whereas most forms of HF are characterized by increased left ventricular (LV) mass, the patterns of ventricular remodeling in HFpEF and HFrEF are distinct. For example, in HFrEF patients the LV chamber dilation is a defining characteristic and in the majority of HFpEF patients ventricular chamber size is normal, or near normal but comes with increased wall thickness.
Treatment
Several strategies to treat patients with heart failure are currently available. The American College of Cardiology (ACC) and the European Society of Cardiology (ESC) regularly publish updated guidelines for treating HFrEF or HFpEF. In addition to medical treatment, guidelines cover catheter-based techniques (e.g. percutaneous coronary interventions, stents, valve repair), the use of implantable cardioverter-defibrillators (ICDs) and cardiac resynchronization therapy (CRT).
While rapid progress to develop HFrEF treatment options has been made over the past 10 years, several studies to establish new treatment options for HFpEF have failed. Thus, treating HFrEF patients is generally more straightforward and recommended medical guideline therapies can reduce mortality and morbidity. None of the available HFpEF treatments have shown to reduce mortality or morbidity in patients, and treatment for HFpEF patients remains empirical and is more challenging. Current guidelines recommend focusing on improving patients’ well-being and, if disease progression cannot be slowed, recommend using a variety of alternatives, including novel devices.
Notwithstanding, even with the best medical care, patients with HFrEF, and more oftenHFpEF, suffer from persistent symptoms and experience poor quality of life (QoL).
Interatrial Shunting
Atrial septostomy (or balloon atrial septostomy, BAS), to create a right-to-left interatrial communication (interatrial shunt), was first described by Rashkind and Miller in 1966. The procedure is regarded as safe and effective, and it remains an important
interventional procedure in the palliation of certain rare congenital heart diseases, including in patients with advanced PAH.
Interatrial shunting to create a left-to-right shunt decompressing the left heart is also being investigated in patients with HF. Elevated LA pressure, particularly during exercise, results in HF symptoms, including exertional dyspnea. Thus, the creation of a small, controlled left-to-right interatrial shunt should result in rapid and potentially sustained improvements of exercise capacity, other symptoms.
The concept of interatrial shunting in HF patients is supported by observations in patients with mitral valve stenosis and a concomitant atrial septal defect (ASD), as well as, from findings in ECMO patients with increased LA pressure. Both groups appear to benefit from BAS, suggesting that interatrial shunting may also improve symptoms in patients with HFpEF or HFrEF.
However, using BAS alone has drawbacks. Spontaneous shunt closure is frequent (data from PAH patients suggest that repeat procedures are needed in 25% or more of patients), and it is difficult to establish a defined shunt diameter with BAS alone.
The Atrial Flow Regulator (AFR)
The AFR is a CE-marked EU Class III implantable, interatrial, shunting device dedicated to addressing shortcomings of atrial septostomy and improving overall safety and outcomes of interatrial shunting in patients with HFpEF or HFrEF.
The device has a two-disc configuration with a central fenestration to maintain an interatrial shunt, and to allow blood to flow through the central fenestration of the AFR, from the left to the right side of the heart following BAS. For HF patients, the AFR is available with two different waist-heights and two different disc sizes, to accommodate different thicknesses of the atrial septal wall and overall cardiac anatomy, respectively. In addition, the device is available with two different fenestration diameters. The diameter of the fenestration is determined by the desired size of interatrial communication using a sizing algorithm.
The AFR is implanted using standard transcatheter techniques via the femoral vein and under imaging. In brief, following transseptal puncture of the interatrial
septum, the puncture site is expanded to the desired shunt diameter using a high pressure
balloon. Thereafter, a transcatheter delivery sheath is used to deliver and implant the AFR using its dedicated pusher cable.
Following the implant of an AFR, an interatrial left to right shunt is created whereby blood from the high-pressure LA flows into the low-pressure RA. To adequately and safely manage the flow and pressure gradients in patients receiving an AFR implant, an algorithm to select adequate AFR fenestration diameters and
sizes has been developed by clinical studies.

Access to the AFR
The AFR is CE-Marked for use in patients with HFpEF or HFrEF and is for sale in Europe and other international markets. The AFR is not approved for use in the United States. Clinical studies to assess the long-term effects of the AFR are ongoing.
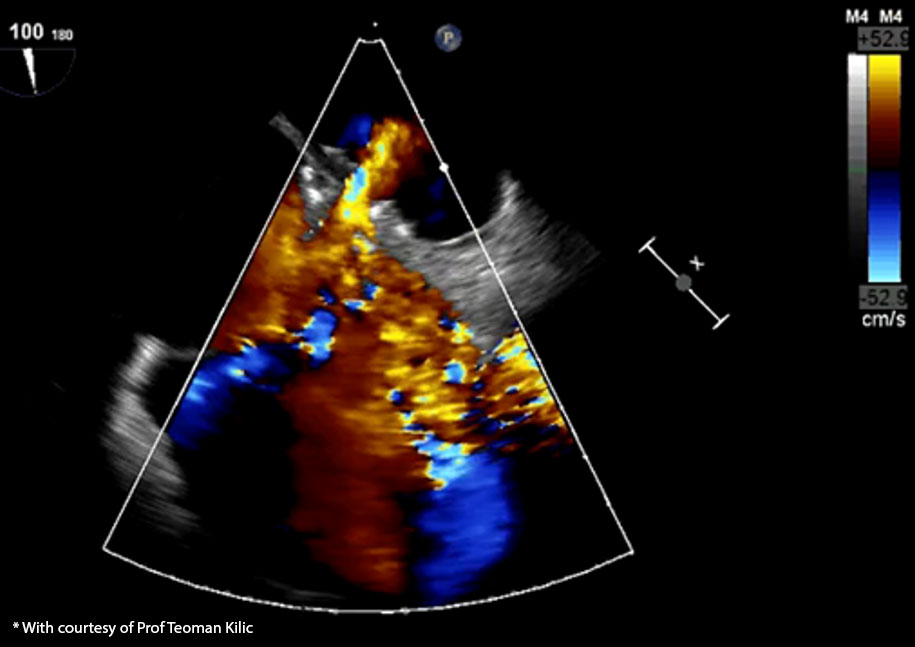
ECHO showing placement of the AFR in the interatrial septum of a patient.*

3D TEE representation showing an implanted AFR*
* Data on file


