
Occlutech ASD Occluder

technical success rate.1,2

efficient closure after
12 months.1, 2

force free angulation
during release.7, 8
Product features and benefits
- The absence of the LA-hub provides flexibility.4
- The absence of the LA-hub may facilitate neo-endothelialization.3, 5
- Comparative study showed that the Occlutech ASD Occluder is associated with shorter procedure time (p= 0.166) and less radiation (p= 0.001).7
- The ball-connector of the Occlutech ASD Occluder provides free rotation and force-free angulation up to 50 degrees and therefore facilitates implantation, alignment with the natural septal angle for operator confidence.6,7
- The Occlutech ASD Occluder shows good procedural outcome and a low complication rate.10

Products are not approved or available in all geographies. Please check with your Occlutech representative for complete information.
Material and design characteristics8
- Optimized braiding pattern and less material at left atrial disc
- Lower pull in forces and improved delivery sheath compatibility

Compatibility chart & ordering information
Over
0
ASD Occluders
delivered in more than 70 countries
Every beat counts
Animation showing in a general way how an Occlutech ASD Occluder is implanted.

3D TEE image of an implanted Occlutech
ASD Occluder.
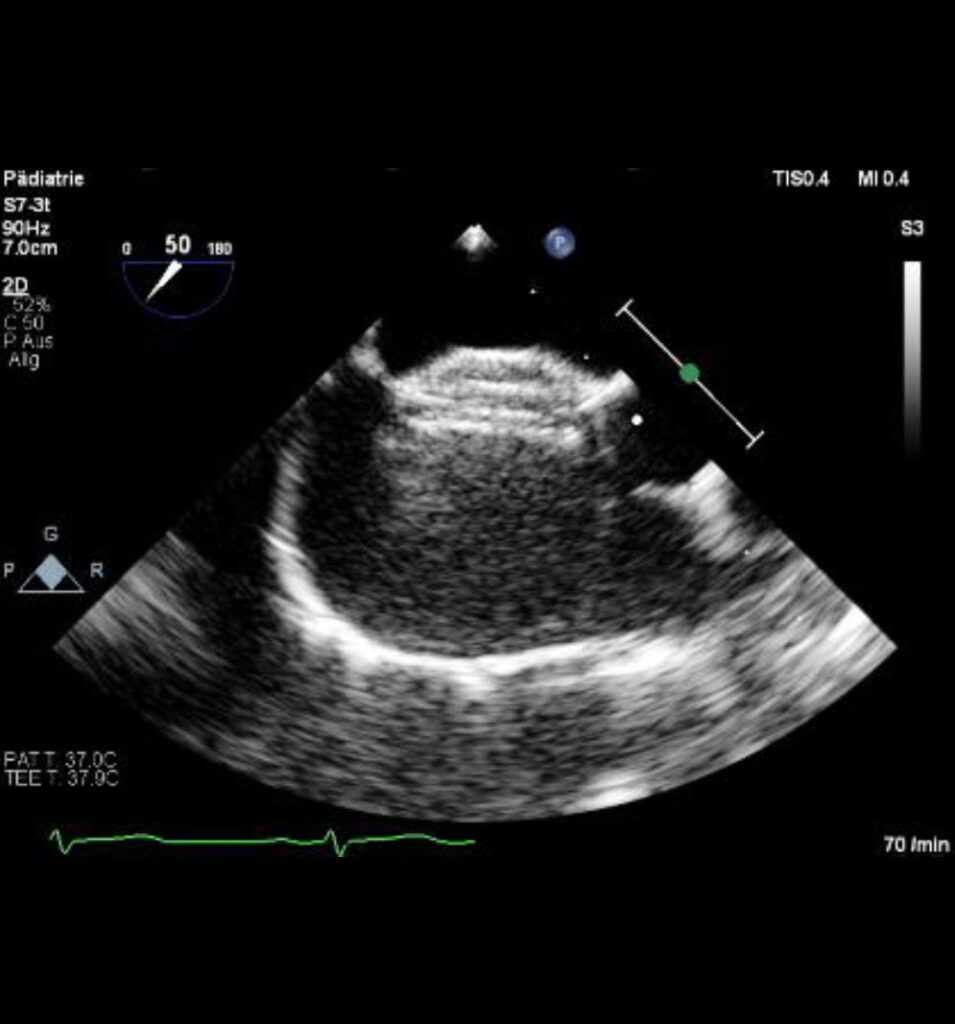
Echo image of an Occlutech ASD Occluder implanted in the Septum.

Sharp angulation between pusher cable and occluder in X-Ray image.
Content references:
- Kenny_A randomized, controlled, multi-center trial of the efficacy and safety of the Occlutech Figulla Flex-II Occluder compared to the Amplatzer Septal Occluder for transcatheter closure of secundum atrial septal defects. Catheter Cardiovasc Interv. 2018;1–6. DOI: 10.1002/ccd.27899
- Nir-David_ASD_IMAJ_Vol 19_September 2017_Comparing the Performance of Amplatzer and Occlutech Figulla Septal Occluders The peadiatric point of view. Doi:19: 557–561
- Haas_Catheterization and Cardiovascular Interventions (2016)_Closure of Secundum Atrial Septal Defects by Using the Occlutech Occluder Devices in More Than 1300 Patients: The IRFACODE Project: A Retrospective Case Series, DOI: 10.1002/ccd.26497
- Tanabe_Journal of the American Heart Association (2021;10:e019282). Angioscopic Evaluation of Atrial Septal Defect Closure Device Neo-Endothelialization DOI: 10.1161/JAHA.120.019282 1
- Verified by pre-clinical studies, data on file (OCC 605-20 Final GLP Study Report)
- Leong_Cardiology in the Young (2022)_The-ceraflex-and-figulla-atrial-septal-occluders-early-and-intermediate-term-safety-and-efficacy-study_doi: 10.1017/S1047951121004728
- Aparisi_INTERVENTIONAL CARDIOLOGY_Cardiology Journal (2020), Vol. 27, No. 5, 524–532_Aparisi_Comparison of Figulla Flex® and Amplatzer™_DOI: 10.5603/CJ.a2020.0058 devices for atrial septal defect closure: A meta-analysis
- Verfied by data on file
- P17F03.030ml.02 ASD IFU data on file
- Leong et al., 20226 Early-onset complication rates in Amplatzer septal occluder, Ceraflex Septal occluder and Figulla atrial septal occluder were 3.83%, 5.56%, and 0%
Not approved or available for sale in the United States.
Product(s) may not be available in all markets because product availability is subject to the regulatory
and/or medical practices in individual markets. Please contact your Occlutech representative if you
have questions about the availability of Occlutech products in your area.


